The Science of Shaved Snow

“Fluffy” isn’t just a feeling — it’s physics.
When you take your first bite of shaved snow, what you’re experiencing isn’t ordinary ice doing something extraordinary. It’s a carefully engineered structure at the microscopic level. The difference between shaved ice and shaved snow is defined by the nucleation rate and crystal symmetry achieved during the freezing process. While traditional shaved ice forms large, disordered crystals, our -50° freezing suppresses grain growth to create a low-density, micro-crystalline lattice. The result is a structurally pure, “dry” snow texture that absorbs flavor without the immediate melting found in coarser ice products.
Fluffy Has a Shape
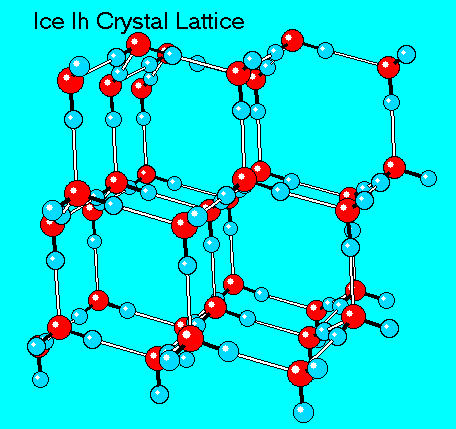
When people describe shaved ice as “crunchy,” they’re unknowingly talking about geometry. Traditional shaved ice is made from Hexagonal Ice Ih, the most common form of ice found in nature. At the molecular level, water molecules arrange themselves into large, orderly hexagonal crystals. This structure is stable, rigid, and loud when you bite into it. When ice is frozen and shaved at standard temperatures, those large crystals stay intact, producing the sharp flakes most people associate with shaved ice. (source)
Why Colder Means Fluffier
Fluffiness isn’t about air — it’s about timing. The texture of ice is determined by how fast crystals form versus how fast they’re allowed to grow. Freezing at −50° dramatically slows crystal growth while allowing many tiny crystals to form at once. Instead of growing into large hexagonal plates, the water locks into a low-density micro-crystalline lattice almost instantly. This creates what scientists call stacking-disordered ice, a mix of cubic and hexagonal layers. That structural disorder is what turns ice from crunchy to cloud-like.
Frozen Mid-Motion
Most ice is allowed to settle into a calm, stable state. Shaved snow never gets that chance. While true amorphous, glass-like ice forms at much colder temperatures, freezing at −50° pushes water far away from equilibrium. The result is a metastable structure that behaves less like a solid block and more like a frozen pause button on a liquid. When shaved, it breaks apart softly instead of fracturing into hard shards, giving snow its signature lightness.
Why Snow Holds Flavor
Smaller crystals create more surface area and more space between molecules. At extreme cold, pure ice can still rearrange itself at the molecular level, creating an unusually active surface. Practically speaking, this means shaved snow acts like a sponge. Flavors don’t run off or pool at the bottom — they’re absorbed into the porous structure. Coffee, chocolate, and fruit infusions soak in evenly, delivering flavor throughout every bite.
Fluff, Explained
Traditional shaved ice is orderly, rigid, and brittle. Shaved snow is intentionally unfinished. By stopping crystal growth before order fully takes over, −50° freezing creates ice that’s dry, light, and fluffy by design. Not softer by accident — softer by science.
Enough of the science though, checkout what our "frozen in motion" can do!




